ABSTRACT - PowerPoint PPT Presentation
Title: ABSTRACT
1
Switching vs. Consistent Triptan Treatment and
Headache-Related Disability Results of the
American Migraine Prevalence and Prevention
(AMPP) Study Dawn C. Buse PhD1 Daniel Serrano
PhD2 Shashi H. Kori MD3 Cedric M. Cunanan MPH4
Aubrey N. Manack PhD4 Michael L. Reed PhD2
Richard B. Lipton MD1 1. Albert Einstein College
of Medicine, Bronx, NY 2. Vedanta Research,
Chapel Hill, NC 3. MAP Pharmaceuticals, Mountain
View, CA 4. Allergan Inc., Irvine, CA
BACKGROUND
RESULTS
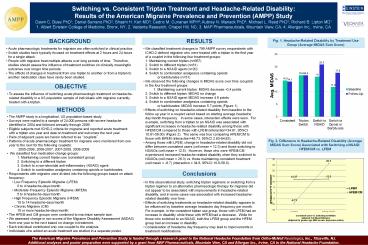
Fig. 1. Headache-Related Disability by Treatment
Use Group (Average MIDAS Sum Score)
- Acute pharmacologic treatments for migraine are
often switched in clinical practice. - Switch studies have typically focused on
treatment effects at 2 hours and 24 hours for a
single attack. - People with migraine treat multiple attacks over
long periods of time. Therefore, studies should
assess the influence of treatment switches on
clinically meaningful outcomes over longer time
periods. - The effects of changes in treatment from one
triptan to another or from a triptan to another
medication class have rarely been studied.
- We classified treatment changes in 799 AMPP
survey respondents with ICHD-2 defined migraine
who were treated with a triptan in the first year
of a couplet in the following four treatment
groups - 1. Maintaining current triptan (n667)
- 2. Switch to different triptan (n81)
- 3. Switch to a NSAID agent (n20)
- 4. Switch to combination analgesics containing
opioids - or barbiturates (n31)
- We observed the following changes in MIDAS score
over time (couplet) in the four treatment groups - 1. Maintaining current triptan MIDAS
decrease -0.4 points - 2. Switch to different triptan MIDAS no change
- 3. Switch to a NSAID agent MIDAS increase 4.9
points - 4. Switch to combination analgesics containing
opioids - or barbiturates MIDAS increase
5.7 points (Figure 1) - Effects of switching on headache-related
disability from baseline to the follow up year in
a couplet varied based on starting average
headache day/month frequency. In some cases,
interaction effects were seen. For example,
switching from a triptan to an NSAID was
associated with significant increases in
headache-related disability among those with
HFEM/CM compared to those with LFEM
(interaction34.81, 95CI 10.61-59.00) (Figure
2). The same was true comparing HFEM/CM to those
with MFEM (interaction48.73, 95CI 2.63-94.83). - Among those with LFEM, change in headache-related
disability did not differ between consistent
users (cell mean 12.3) and those switching to
NSAIDs (cell mean 12.5). However, those who
were HFEM/CM experienced increased
headache-related disability when they switched to
NSAIDs (cell mean 26.3) vs. those maintaining
consistent treatment (cell mean -8.7)
(interaction 34.8, 95CI 10.6-59.0).
OBJECTIVE
- To assess the influence of switching acute
pharmacologic treatment on headache-related
disability in a US population sample of
individuals with migraine currently treated with
a triptan.
METHODS
- The AMPP study is a longitudinal, US
population-based study. - Surveys were mailed to a sample of 24,000 persons
with severe headache identified in 2004 and
followed annually through 2009. - Eligible subjects met ICHD-2 criteria for
migraine and reported acute treatment with a
triptan one year and data on treatment and
outcomes the next year. - Pairs of adjacent years are herein referred to as
couplets. - Patterns of acute pharmacologic treatment for
migraine were monitored from one year to the next
for the following couplets - 2005-2006, 2006-2007, 2007-2008,
2008-2009 - We classified four medication-change groups
- 1. Maintaining current triptan use (consistent
group) - 2. Switching to a different triptan
- 3. Switch to a non-steroidal anti-inflammatory
(NSAID) agent - 4. Switch to combination analgesics containing
opioids or barbiturates - Respondents with migraine were divided into the
following groups based on attack frequency - Low Frequency Episodic Migraine (LFEM)
- 0 to 4 headache-days/month
- Moderate Frequency Episodic Migraine (MFEM)
- 5 to 9 headache-days/month
- High Frequency Episodic Migraine (HFEM)
- 10 to 14 headache-days/month
- Chronic Migraine (CM)
Fig. 2. Difference in Headache-Related Disability
(Average MIDAS Sum Score) Associated with
Switching a NSAID HFEM/CM vs. LFEM
Conclusions
- In this observational study, switching triptan
regimens or switching from a triptan regimen to
an alternative pharmacologic therapy for migraine
did not appear to be associated with improvements
in headache-related disability, and in some cases
was associated with increased headache-related
disability over time. - Effects of switching treatments on
headache-related disability appears to be
influenced by baseline average headache day
frequency per month. For example, in the
consistent triptan use group, those with LFEM had
an increase in disability while those with HFEM
had a decrease. While for those who switched to
an NSAID, both the LFEM group and the HFEM group
had an increase in disability. - Consideration of headache day frequency may lead
to improvements in treatment modifications.
The American Migraine Prevalence and Prevention
Study is funded through a research grant to the
National Headache Foundation from Ortho-McNeil
Neurologics, Inc., Titusville, NJ. Additional
analyses and poster preparation were supported by
a grant from MAP Pharmaceuticals, Mountain View,
CA and Allergan Inc., Irvine, CA to the National
Headache Foundation.