Mass Spectrometry - PowerPoint PPT Presentation
1 / 26
Title:
Mass Spectrometry
Description:
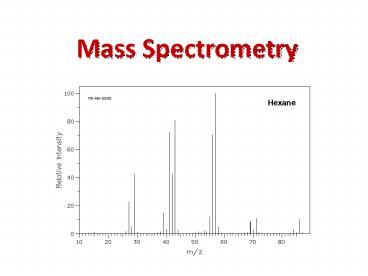
The cations are detected by the MS instrumentation. Mass Spec - Introduction ... Mass Spec - Instrumentation. Carey, Chapter 13. An Example Hexane (mw = 86) M ... – PowerPoint PPT presentation
Number of Views:66
Avg rating:3.0/5.0
Title: Mass Spectrometry
1
Mass Spectrometry
Hexane
2
Prelab and Datasheet
- Reading
- Technique P in lab text, p. 160-171
- Carey, Chapter 13 (4 pages at end of chapter)
- Notebook nothing
- Datasheet
- Handed out in class
- Due at end of lab period (so come prepared and
ready to go to work).
3
Mass Spec - Introduction
- Very different from IR and NMR
- Absorption of electromagnetic energy
- Sample can be recovered and reused
- Mass spectrometry
- Records what happens when an organic molecule is
hit by a beam of high-energy electrons - Sample is completely destroyed
4
Mass Spec - Introduction
- What does a mass spectrum tell us?
- Molecular weight
- Molecular formula
- Either directly or in conjunction with other
kinds of spectra such as IR or NMR - Fragmentation pattern
- Key pieces of what the molecule looks like (such
as methyl, ethyl, phenyl, or benzyl groups
5
Mass Spec - Introduction
- We hit an organic molecule with a beam of
electrons (usually 70-75 eV) - That removes an electron from the molecule
resulting in the molecular ion (a radical cation) - The molecular ion then fragments in smaller
radicals and cations - The cations are detected by the MS instrumentation
6
Mass Spec - Introduction
7
Mass Spec - Instrumentation
Sample Introduction
Data Output
Data Systems
Inlet
Mass Analyzer
Ion Source
Ion Detector
High vacuum
8
Mass Spec - Instrumentation
Carey, Chapter 13
9
An Example Hexane (mw 86)
57
Base peak (always 100)
43
Molecular ion peak
Various cation fragments
29
71
M
Molecules with 13C
10
Hexane Fragmentation
CH3 - CH2 - CH2 - CH2 - CH2 - CH3
M 86
71
57
43
29
15
11
Fragmentation Guidelines
- Cleavage generally favors the formation of the
most stable cations and radicals - tertiary gt secondary gt primary gt methyl
- resonance stabilized cations often formed
- small, neutral molecules often eliminated (water,
carbon monoxide, ammonia, ketene) - Also depends upon the relative bond strengths of
the bonds being broken
12
Common Fragment Ions
More in text, p. 169, Table 2P-2.
13
Isotopic Clusters
- Especially useful for identifying chlorine and
bromine - Chlorine has two isotopes 35Cl and 37Cl in about
a 75/25 ratio (avg MW 35.45) - So compounds that have a chlorine will have 35Cl
(75) and 37Cl (25) - The molecular ion peak will show as two peaks in
a 75/25 ratio, 2 mass units apart
14
M 112
Chlorobenzene (MW 112)
77 (phenyl)
M2 114
15
Isotopic Clusters
- Bromine also has two isotopes
- 79Br and 81Br (avg MW 79.9)
- Molecules that contain bromine have a 50/50
chance of getting one or the other - Molecular ions of compounds containing a single
bromine will have two peaks, 2 mass units apart,
of about the same relative intensity
16
Bromobenzene
Base peak at 77 (phenyl)
M 156
M2 158
17
More Things to Look For
- Iodine does not have an isotope cluster, but
often shows a major peak at M-127 due to loss of
I - Alcohols will often have a peak at M-18 (loss of
water) - Nitrogen rule if a compound has an odd number
of nitrogens, the MW is odd (and M) - If the number of nitrogens is even (or zero), the
MW will be even - Many compounds do not show a good M peak
- Alcohols, highly branched compounds
18
1-iodobutane (MW 184)
57 (184-127)
M 184
19
45 (Loss of methyl from M)
2-propanol (MW 60) Alcohols often do not
show an M peak. M-1 is common, but may also be
missing
M-1 59
20
M-18 56
1-butanol
M 74
21
77 base peak (phenyl)
Benzamide (MW 121)
M 121
105 (Loss of NH2)
22
M-17 43
M-15 45
M 60
Acetic Acid
23
M 106
Benzaldehyde (aldehydes often have a large
M-1 peak)
77
24
Interpreting Spectra
- If an IR is present, examine for functional
groups - If an NMR is present
- Validate IR functional groups via chemical shift
data - Look for characteristic shifts and splitting
patterns - Examine the mass spec
- Look for the presence of Cl, Br, I, and N
- Determine the MW of the compound from M
- Look for common fragments and patterns
- Subtract known atoms from M to determine C and H
- Postulate a molecular formula
- Postulate a structure consistent with the known
data
25
What is this compound?
M 122
26
What is this compound?
M 86