Principles%20of%20Bioinorganic%20Chemistry%20-%202003 - PowerPoint PPT Presentation
Title:
Principles%20of%20Bioinorganic%20Chemistry%20-%202003
Description:
The grade for this course will be determined by a term exam (35%), a written ... Labile metals used (nature works at a kilohertz) ... – PowerPoint PPT presentation
Number of Views:63
Avg rating:3.0/5.0
Title: Principles%20of%20Bioinorganic%20Chemistry%20-%202003
1
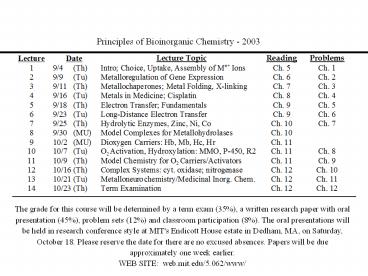
Principles of Bioinorganic Chemistry - 2003
The grade for this course will be determined by a
term exam (35), a written research paper with
oral presentation (45), problem sets (12) and
classroom participation (8). The oral
presentations will be held in research conference
style at MIT's Endicott House estate in Dedham,
MA, on Saturday, October 18. Please reserve the
date for there are no excused absences. Papers
will be due approximately one week earlier. WEB
SITE web.mit.edu/5.062/www/
2
Principles of Bioinorganic Chemistry
Two Main Avenues of Study
- Understand the roles of naturally occurring
inorganic elements in biology. By weight, gt 50
of living matter is inorganic. Metal ions at the
core of biomolecules control many key life
processes. - Use metals as probes and drugs
Examples Cisplatin, auranofin as
pharmaceuticals Cardiolyte (99mTc)
and Gd, imaging agents MoS42-, Wilsons disease
cancer??
3
Respiration - Three O2 Carriers in Biology
oxyHb, Mb
deoxyHb, Mb
oxyHc
deoxyHc
deoxyHr
oxyHr
4
The Heme Group the Defining Example of a
Bioinorganic Chip
Peripheral carboxylates and axial ligands matter!
5
The Major Metal Units in ET Proteins
Iron-Sulfur clusters, electron transfer relay
stations
6
(No Transcript)
7
Structure of the Streptomyces lividans (KcsA)
Potassium Channel (MacKinnon, et al., 1998)
Extracellular
Top view
Cytoplasm
8
Cobalamin structures
9
Three Inorganic Compounds Used in Modern Medicine
10
Course Organization
- What metals? How taken up? How assemble?
- How do cells regulate metal ion concentrations?
Homeostasis. - How do metal ions fold biopolymers?
- How is the correct metal ion inserted into its
site? - Electron transfer metalloproteins.
- Substrate binding and activation, non-redox.
- Atom and group transfer (main oxygen chemistry).
- Protein tuning of active sites.
11
Choice, Uptake and Assembly of Metal Ions in
Cells
PRINCIPLES
- Relatively abundant metal ions used
(geosphere/biosphere) - Labile metals used (nature works at a kilohertz)
- Low abundance metals concentrated by ATP driven
processes - Entry to the cell controlled by specific channels
and pumps - Co-factors employed bioinorganic chips
(porphyrins) - Self-assembling units form - from geosphere
- Metallochaperones assure that metal ions find
their proteins
ILLUSTRATIONS
- The selectivity filter of the potassium channel
- Uptake of iron
12
Relative abundance of metal ions in the earths
crust and seawater
13
Iron Uptake in the Cell
The Challenge
- Iron is the second most abundant metal after
aluminum - Its Fe(II) and Fe(III) redox states render it
functionally useful - At pH 7, iron is insoluble (10-18 M)
- The challenge How to mobilize iron in the
biosphere?
The Solutions In bacteria, siderophores In
humans, transferrin
14
Synthesis and Structure of Dinuclear Ferric
Citrate Complexes
It will be interesting to determine whether
solutions of 1 or 2 are taken up by living cells.
Shweky et. al. Inorg. Chem. 1994, 33, 5161-5162.
15
Ferric Citrate-Binding Site of Outer Membrane
Transporter FecA
Ferguson et. al. Science, 2002, 295, 1715-1719.
16
Diiron Core of the Outer Membrane Transporter FecA
2.02 Å
1.98 Å
2.02 Å
2.00 Å
2.01 Å
Fe
Fe
2.05 Å
2.00 Å
1.96 Å
2.00 Å
2.01 Å
17
Enterobactin a Bacterial Siderophore
18
Enterobactin, a Cyclic Triserine Lactone
A specific cell membrane receptor exists for
ferric enterobactin. Release in the cell can
occur by hydrolysis of the lactone, reduction to
Fe(II), and/or lowering the pH.
19
Structure of Vanadium(IV) Enterobactin
20
Scheme showing the ATP-driven uptake of ferric
enterobactin into E. coli cells through a
specific receptor in the cell membrane.
Does not distinguish D from L
outer membrane
cytoplasmin membrane
intracellular esterase hydrolyzes Ent, releases
iron
See Raymond, Dertz, and Kim, PNAS, 100, 3584.