In Antarctica the thermometer reads
Title:
In Antarctica the thermometer reads
Description:
Physics 1710 Warm-up Quiz 0 In Antarctica the thermometer reads 20 C; what is this temperature on a Fahrenheit thermometer? -36 oF-11 oF –
Number of Views:203
Avg rating:3.0/5.0
Title: In Antarctica the thermometer reads
1
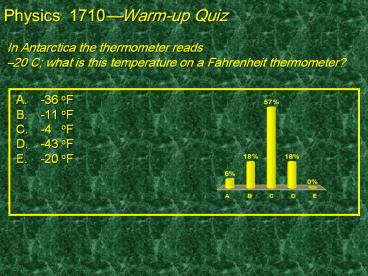
In Antarctica the thermometer reads 20 C what
is this temperature on a Fahrenheit thermometer?
Physics 1710Warm-up Quiz
0
- -36 oF
- -11 oF
- -4 oF
- -43 oF
- -20 oF
121 122 123 124 125 126 127 128 129 130 131 132 133 134 135 136 137 138 139 140
2
Physics 1710 Chapter 20 Heat 1st Law of Thermo
0
- Solution
- oF (180 oF/100 oC) oC 32oF
- (1.8 oF/oC)(-20 oC) 32oF
- (-36 32) oF
- -4oF
3
Physics 1710 Chapter 20 Heat 1st Law of Thermo
0
What will happen to a heated ring? The hole
will(1) get larger (2) get smaller (3) stay
the same.
Peer Instruction Time
4
What will happen to a heated ring? The hole will
Physics 1710 e-Quiz
0
- Get larger
- Get smaller
- Stay the same
41 42 43 44 45 46 47 48 49 50 51 52 53 54 55 56 57 58 59 60
61 62 63 64 65 66 67 68 69 70 71 72 73 74 75 76 77 78 79 80
5
Physics 1710 Chapter 20 Heat 1st Law of Thermo
0
- Time for Real Physics!
6
Physics 1710 Chapter 20 Heat 1st Law of Thermo
0
- Why did it happen?
?L
?L
?L a L?T
7
Physics 1710Chapter 19 Temperature
0
- Ideal Gas Law
- Idealizations
- no interaction between atoms
- no volume occupied by atoms
- Number of atoms n m/M,
- m mass M molar mass
- PV n R T
- R 8.315 J/ mol K
- 0.08214 L?atm/mol K
- 22.4 L?atm /273.16 mol K
8
Physics 1710Chapter 19 Temperature
0
- Boltzmann Constant
- k R/NA
- k 1.38 x 10 -23 J/K 1 yJ/ 7.25 K
- 1 eV / 11,600 K
- PV N kT
9
Physics 1710Chapter 19 Temperature
0
- Summary
- Temperature is a measure of the average kinetic
energy of a system of particles. - Thermal Equilibrium means that two bodies are at
the same temperature. - The Zeroth Law of Thermodynamics states that
if system A and B are n thermal equilibrium with
system C, then A and B are in thermal Equilibrium
with each other.
10
Physics 1710Chapter 19 Temperature
0
- Kelvin is a unit of temperature where one degree
K is 1/279.16 of the temperature of the triple
point of water (near freezing). - TC (100/180) (TF 32 F)
- TF (180/100) TC 32 F
- ?L/L a?T
- PV n R T N kT
11
Physics 1710 Chapter 20 Heat 1st Law of Thermo
0
- 1' Lecture
- The internal energy is the total average energy
of the atoms of an object, average kinetic plus
average potential. - Heat is the change in internal energy.
- The change in temperature is proportional to the
change in internal energy (heat flow) when there
is no change of phase and the system does no
work. - The first law of thermodynamics states
- ?E ?Q - W
12
Physics 1710 Chapter 20 Heat 1st Law of Thermo
0
- 1' Lecture
- Conduction is the flow of kinetic energy from
atom to atom. - Convection is the transport of energy by bulk
motion of atoms. - Radiation is the transfer of energy by
electromagnetic waves.
13
Physics 1710 Chapter 20 Heat 1st Law of Thermo
0
- Heat
- Heat is the macroscopic manifestation of
microscopic internal energy. - Heat ?Q in calories (or BTU)
- 1 calorie (cal) is the amount of energy required
to raise the temperature of 0.001 kg of water
from 14.5 C to 15.5 C (?T 1.00 C). - 1 BTU is the heat to raise 1 Lb by 1 F.
- James Prescott Joule (1818-1889) showed
- 1 calorie of heat 4.186 Joule
- 1 J 1 N?m
14
Physics 1710 Chapter 20 Heat 1st Law of Thermo
0
- 1 Calorie 1000 calorie 1 kcal
15
Physics 1710 Chapter 20 Heat 1st Law of Thermo
0
- Heat Capacity
- ?Q C ?T
- C dQ/dT
- Specific Heat
- c C /m
- C (1/m)dQ/dT
- ?Q (mc)?T
16
How much heat is required to raise the
temperature of 1 kg of water (1 liter) from 20 C
to 100 C? Recall c 1.00 kcal/kgC?
Physics 1710 e-Quiz
0
- 1.0 kcal
- 20 kcal
- 80 kcal
- 100 kcal
- None of the above.
1 2 3 4 5 6 7 8 9 10 11 12 13 14 15 16 17 18 19 20
21 22 23 24 25 26 27 28 29 30 31 32 33 34 35 36 37 38 39 40
17
Physics 1710 Chapter 20 Heat 1st Law of Thermo
0
- Guided practice
- ?Q (mc)?T
- How much heat is required to raise the
temperature of 1 kg of water (1 liter) from 20 C
to 100 C? Recall c 1.00 kcal/kgC?
?Q (mc)?T ?Q (1.00 kg)(1.00 kcal/kg C) (100.
C- 20. C) ?Q 80. kcal 80.kcal? 4.186 J/cal
396. kJ
18
Physics 1710 Chapter 20 Heat 1st Law of Thermo
0
- Change of Phase and Latent Heat
- It requires an energy investment to change the
phase from solid to liquid to gas. - By breaking the bonds that hold atoms, they can
have the same kinetic energy but different total
energies. - The energy to change the phase is hidden and
therefore called latent heat.
19
Physics 1710 Chapter 20 Heat 1st Law of Thermo
0
- Latent heat
- ?Q mL
- For Water
- Fusion (and melting)
- Lf 333 kJ/kg 79.4 kcal/kg
- Vaporization
- Lv 2260 kJ/kg 540 kcal/kg
20
Physics 1710 Chapter 20 Heat 1st Law of Thermo
0
- Work from a Heat Reservoir
- The work done by a system is equal to the loss
of the internal energy. - For an ideal gas
- W ?V1V2 PdV
21
Physics 1710 Chapter 20 Heat 1st Law of Thermo
0
- First Law of Thermodynamics
- ?E ?Q -W
22
Physics 1710 Chapter 20 Heat 1st Law of Thermo
0
- ApplicationsAdiabatic (?Q 0)
- ?E -W
- Isovolumetric (?V 0)
- ?E ?Q
- Isothermal (?T 0)
- W ?V1V2 PdV ?V iV f (nRT/V) dVW n R T
ln(Vf /Vi )
23
Physics 1710 Chapter 20 Heat 1st Law of Thermo
0
- Mid Chapter Summary
- The internal energy is the total average energy
of the atoms of an object. - Heat is the change in internal energy.
- The change in temperature is proportional to the
change in internal energy (heat flow) when there
is no change of phase and the system does no
work. - The first law of thermodynamics states
- ?E ?Q - W
24
Physics 1710 Chapter 20 Heat 1st Law of Thermo
0
- Conduction
- P kA dT/dx
- Examples
- Thermos bottles
- Blankets
- Double pane windows
- Newtons law of cooling P h A(T 2 T1)
- Pans
- R factor or R value
- P A(T 2 T1)/?i Ri
25
Physics 1710 Chapter 20 Heat 1st Law of Thermo
- Convection
- Heat transfer by material transfer
- Forced convection (fluids)
- External force produces material transfer
- Natural Convection
- Buoyancy-driven flow
- Newtons law of cooling applied
- P h A(T 2 T1)
- h depends on flow conditions
26
Physics 1710 Chapter 20 Heat 1st Law of Thermo
- Radiation
- Stefan-Boltzmann Law
- P es AT4
- Wiens Law
- P ?T4
- s 5.6696 x 10-8 W/m2?K4
- Emissivity 0lt e lt1 e ½
- Reflectivity (albedo) R (1- e)
- Energy balance
- P in - es A(Tave ) 4 0
27
Physics 1710 Chapter 20 Heat 1st Law of Thermo
- Global Warming?
- P in ( 1- e?) Psun
- Tave ( 1- e?) Psun /(eGH sA)1/4
- Must understand every parameter
- to be accurate.
28
Physics 1710 Chapter 20 Heat 1st Law of Thermo
- Summary
- Heat is transferred by
- Conductionenergy diffusion
- Convectionmass transport
- Radiationelectromagnetic waves
29
Physics 1710 Chapter 20 Heat 1st Law of Thermo
- Guided Practice
- If the power per unit area (intensity) of
sunlight on the earth is 1.0 kW/m2 and the
emissivity is 0.6, what is the expected avearge
temperatue of the earth? Comment on the effect
of a change in the emissivity or solar
radiation.
P in ?0 2p?0 p/2 I cos ? sin ? r 2d? df P in I
(2pr 2) (½ ) sin2 ? 0 p/2 I (pr 2) 1.0
kW/m2 (3.14)(6.4x106 m)2 1.3 x 1017 W P in es
A(Tave ) 4 Tave P in / es A1/4 1.3 x
1017 /(0.6?5.66 x10-8? 5.1x1014) 1/4 294 K21C
30
Physics 1710 Chapter 20 Heat 1st Law of Thermo
- Guided Practice
- If the power per unit area (intensity) of
sunlight on the earth is 1.0 kW/m2 and the
emissivity is 0.6, what is the expected average
temperature of the earth? Comment on the effect
of a change in the emissivity or solar radiation.