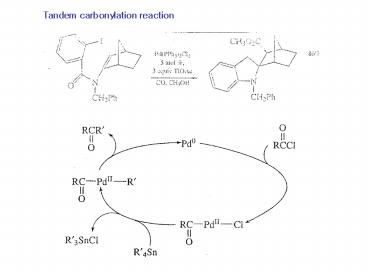
Tandem%20carbonylation%20reaction - PowerPoint PPT Presentation
Title:
Tandem%20carbonylation%20reaction
Description:
Reagents that are relatively strong oxidizing ... oxidize. Existing substituent groups such as CH3, OCH3, and NMe3 exert a directive ... – PowerPoint PPT presentation
Number of Views:136
Avg rating:3.0/5.0
Title: Tandem%20carbonylation%20reaction
1
Tandem carbonylation reaction
2
(No Transcript)
3
Monsanto Process (Acetic acid Synthesis)
150-200oc, 1-40 atm Ref BASF process
cobalt-based high pressure process (200-250oC,
500-700 atm)
4
8.3 Reactions Involving Organonickel Compounds
Allylic halides react with nickel carbonyl,
Ni(CO)4 to give p-allyl complexes.
5
These reactions are believed to involve Ni(I) and
Ni(III) intermediates in a chain process which is
initiated by formation of a small amount of a
Ni(I) sepcies.
6
This couplig reaction has been used
intramolecularly to bring about cyclization of
bis-allylic halides and was found useful in the
preparation of large rings.
Nickel carbonyl is an extremely toxic compound,
and a number of other nickel reagents with
generally similar reactivity can be used in its
place.
7
Mediun sized ring can be formed in intramolecular
reaction.
8
The key aspects of the mechanism are (1) the
reductive elimination which occurs via a diaryl
Ni(III) intermediates and (2) the oxidative
addition which involves a Ni(I) species.
9
A soluble bis-phosphine complexes, Ni(dppe)2Cl2,
is a particularly effective catalyst. The main
distinction between this reaction and
Pd-catalyzed cross coupling is that the nickel
reaction can be more readily applied to
saturated alkyl groups because of a reduced
tendency for b-elimination.
The synthesis of cyclophane-type structures by
use of dihaloarenes and Grignard reagents from
a,w-dihalides.
10
When secondary Grignard reagents are used, the
coupling product sometimes is derived from the
corresponding primary alkyl group. This
transformation can occur by reversible formation.
Styrene serves to stabilize the active catalytic
species, and among the styrene derivatives,
m-trifluoromethylstyrene was the best.
11
The main advantage of nickel is that it reacts
more readily with arylchlorides
and methanesulfonates than does the Pd system.
These reactants may be more economical than
iodides or triflates in large-scale synthesis.
Vinyl phosphates can be used, and these are in
some cases more readily obtained and handled than
vinyl triflates.
12
8.4 Reactions Involving Rhodium and Cobalt
Hydroformylation
The key steps in reaction are addition of
hydridorhodium to the double bond of the alkene
and migration of the alkyl group to the complexed
carbon monoxide.
13
The acylrhodium intermediate is trapped by
internal nucleophiles.
Fischer-Tropsch Process reductive conversion of
carbon monoxide to alkane by reacting with
hydrogen gas. Synthetic hydrocarbon
fuels.(1923-1925) In1944, 600,000 ton/yr was
produced. Since 1957 South Africa use this
method, Sasol Process.
14
Under appropriate conditions, rhodium catalyst
can be used for the decarbonylation of aldehyde
and acyl chlorides.
15
The use of cobalt for synthetic purpose is quite
limited. Vinyl bromides and idodides couple with
Grignard reagents in good yields, but a good
donor solvent such as NMP or DMPU is required as
a cocatalyst.
16
8.5 Organometallic Compounds with p bonding
Among the classes of organic compounds that serve
as p ligands are alkene, allyl, dienes,
cyclopentadiene anion, and aromatic compounds.
The reactivity depends on the following factors
- The number of electrons that can be accommodated
by the metal orbitals - the oxidation level of the metal,
- the electron character of other ligands on the
metal
17
Both thermal and photochemical reactions are used.
P-allyl complexes of nickel can be prepared
either by oxidative addition on Ni(0) or by
transmetallation of a Ni(II) salt.
18
Oxidative decomposition
Trapping experiments
In 1956, Longuet and Orgel propose the complex
compound. In 1959, Criegee isolated the complex.
19
One of the best known of the p-organometallic
compounds is ferrocene.
The molecules behave as an electron-rich aromatic
system, and electrophilic substitution reactions
occur readily. Reagents that are relatively
strong oxidizing agents, such as the halogens,
effect oxidation at iron and destroy the compound.
Effective Atomic Number 18
20
One of the most useful types of p-complexes of
aromatic compounds from the synthetic point of
view are chromium complexes obtained by heating
benzene or other aromatics with Cr(CO)6.
The Cr(CO)3 unit is strongly electron-withdrawing
and activates the ring to nucleophilic attack.
21
oxidize
Existing substituent groups such as CH3, OCH3,
and NMe3 exert a directive effect, often
resulting in a major amount of the meta
substitution product.
22
(No Transcript)
23
(No Transcript)
24
(No Transcript)