Petrochemical Processes - PowerPoint PPT Presentation
Title:
Petrochemical Processes
Description:
As with catalytic cracking, the main reactions occur by carbonium ion and beta scission, yielding two fragments that could be hydrogenated on the catalyst surface. – PowerPoint PPT presentation
Number of Views:2156
Avg rating:3.0/5.0
Title: Petrochemical Processes
1
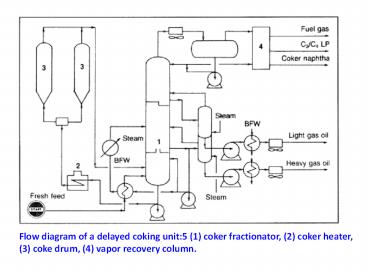
Flow diagram of a delayed coking unit5 (1) coker
fractionator, (2) coker heater, (3) coke drum,
(4) vapor recovery column.
2
(No Transcript)
3
Fluid Coking
- Heated by the produced coke
- Cracking reactions occur inside the heater and
the fluidized-bed reactor. - The fluid coke is partially formed in the heater.
- Hot coke slurry from the heater is recycled to
the fluid reactor to provide the heat required
for the cracking reactions. - Fluid coke is formed by spraying the hot feed on
the already-formed coke particles. Reactor
temperature - is about 520C, and the conversion into coke is
immediate, with complete disorientation of the
crystallites of product coke. - The burning process in fluid coking tends to
concentrate the metals, but it does not reduce
the sulfur content of the coke.
4
- Characteristics of fluid coke
- high sulfur content,
- low volatility, poor crystalline structure, and
low grindability index. - Flexicoking, integrates fluid coking with coke
gasification. - Most of the coke is gasified. Flexicoking
gasification produces a substantial concentration
of the metals in the coke product.
5
Flow diagram of an Exxon flexicoking unit5 (1)
reactor, (2) scrubber, (3) heater, (4) gasifier,
(5) coke fines removal, (6) H2S removal.
6
CATALYTIC CONVERSION PROCESSES
- Catalytic Reforming
- To improve the octane number of a naphtha.
- Aromatics and branched paraffins have high
octane ratings than paraffins and cycloparaffins. - Many reactions e.g. dehydrogenation of
naphthenes and the dehydrocyclization of
paraffins to aromatics. - Catalytic reforming is the key process for
obtaining benzene, toluene, and xylenes (BTX). - These aromatics are important intermediates for
the production of many chemicals.
7
Reformer Feeds
- heavy naphtha fraction produced from atmospheric
distillation units. - Naphtha from other sources such as those produced
from cracking and delayed coking may also be
used. - Before using naphtha as feed for a catalytic
reforming unit, it must be hydrotreated to
saturate the olefins and to hydrodesulfurize and
hydrodenitrogenate sulfur and nitrogen compounds.
- Olefinic compounds are undesirable because they
are precursors for coke, which deactivates the
catalyst. - Sulfur and nitrogen compounds poison the
reforming catalyst. - The reducing atmosphere in catalytic reforming
promotes forming of hydrogen sulfide and ammonia.
Ammonia reduces the acid sites of the catalyst,
while platinum becomes sulfided with H2S.
8
- Important is
- Types of hydrocarbons in the feed.
- Naphthene content
- The boiling range of the feeds
- Feeds with higher end points (200C) are
favorable because some of the long-chain
molecules are hydrocracked to molecules in the
gasoline range. These molecules can isomerize
and dehydrocyclize to branched paraffins and to
aromatics, respectively.
9
Reforming Catalysts
- Bi-functional to provide two types of catalytic
sites, hydrogenation-dehydrogenation sites and
acid sites. - platinum, is the best known hydrogenation-dehydrog
enation catalyst - Alumina, (acid sites) promote carbonium ion
formation - The two types of sites are necessary for
aromatization and isomerization reactions.
10
Reforming Reactions
- Pt/Re catalysts are very stable, active, and
selective. - Trimetallic catalysts of noble metal alloys are
also used for the same purpose. - The increased stability of these catalysts
allowed operation at lower pressures.
Reforming Catalysts
Aromatization
11
- The reaction is endothermic i.e. favoured _at_
higher temp and lower pressures. - Effect of temp on the conversion and selectivity
12
Catalytic Cracking
- Catalytic cracking (Cat-cracking) To crack
lower-value stocks and produce higher-value light
and middle distillates. - To produce light hydrocarbon gases, which are
important feedstocks for petrochemicals. - To produce more gasoline of higher octane than
thermal cracking. This is due to the effect of
the catalyst, which promotes isomerization and
dehydrocyclization reactions. - Feeds vary from gas oils to crude residues
- Polycyclic aromatics and asphaltenes peoduce
coke.
13
Catalytic Catalysts
- Acid-treated clays were the first catalysts used.
- Replaced by synthetic amorphous silica-alumina,
which is more active and stable. - Incorporating zeolites (crystalline
alumina-silica) with the silica/alumina catalyst
improves selectivity towards aromatics. These
catalysts have both Lewis and Bronsted acid sites
that promote carbonium ion formation. An
important structural feature of zeolites is the
presence of holes in the crystal lattice, which
are formed by the silica-alumina tetrahedra. Each
tetrahedron is made of four oxygen anions with
either an aluminum or a silicon cation in the
center. Each oxygen anion with a (II) oxidation
state is shared between either two silicon, two
aluminum, or an aluminum and a silicon cation.
14
Catalytic Catalysts
Bronsted acid sites in HY-zeolites mainly
originate from protons that neutralize the
alumina tetrahedra. When HY-zeolite (X- and
Y-zeolites are cracking catalysts ) is heated to
temperatures in the range of 400500C, Lewis
acid sites are formed.
15
Zeolite Catalysts
- Highly selective due to its smaller pores, which
allow diffusion of only smaller molecules through
their pores, and to the higher rate of hydrogen
transfer reactions. However, the silica-alumina
matrix has the ability to crack larger molecules.
- Deactivation of zeolite catalysts occurs due to
coke formation and to poisoning by heavy metals. - Deactivation may be reversible or irreversible.
- Reversible deactivation occurs due to coke
deposition. This is reversed by burning coke in
the regenerator. - Irreversible deactivation results as a
combination of four separate but interrelated
mechanisms zeolite dealumination, - zeolite decomposition, matrix surface collapse,
and contamination by metals such as vanadium and
sodium.
16
Cracking Reactions
- A major difference between thermal and catalytic
cracking is that reactions through catalytic
cracking occur via carbocation intermediate,
compared to the free radical intermediate in
thermal cracking. - Carbocations are longer lived and accordingly
more selective than free radicals. - Acid catalysts such as amorphous silica-alumina
and crystalline zeolites promote the formation of
carbocations. The following illustrates the
different ways by which carbocations may be
generated in the reactor
17
(No Transcript)
18
(No Transcript)
19
(No Transcript)
20
Aromatization Reactions
- Dehydrocyclization reaction. Olefinic compounds
formed by the beta scission can form a
carbocation intermediate with the configuration
conducive to cyclization.
Once cyclization has occurred, the formed
carbocation can lose a proton, and a cyclohexene
derivative is obtained. This reaction is aided by
the presence of an olefin in the vicinity
(RCHCH2).
21
(No Transcript)
22
Cracking Process
- Most catalytic cracking reactors are either fluid
bed or moving bed. - In FCC, the catalyst is an extremely porous
powder with an average particle size of 60
microns. - Catalyst size is important, because it acts as a
liquid with the reacting hydrocarbon mixture. - In the process, the preheated feed enters the
reactor section with hot regenerated catalyst
through one or more risers where cracking occurs.
A riser is a fluidized bed where a concurrent
upward flow of the reactant gases and the
catalyst particles occurs.
23
- The reactor temperature is usually held at about
450520C, and the pressure is approximately
1020 psig. - Gases leave the reactor through cyclones to
remove the powdered catalyst, and pass to a
fractionator for separation of the product
streams. Catalyst regeneration occurs by
combusting carbon - deposits to carbon dioxide and the regenerated
catalyst is then returned
24
Typical FCC reactor/regenerator
25
Isomerization
- Reactions leading to skeltal rearrangements over
Pt catalysts
26
Hydrocracking
- A hydrogen-consuming reaction that leads to
higher gas production
Hydrdealkylation
A cracking reaction of an aromatic side chain in
presence of hydrogen
27
Deep Catalytic Cracking
- Deep catalytic cracking (DCC) is a catalytic
cracking process which selectively cracks a wide
variety of feedstocks into light olefins. - It produces more olefines than FCC.
28
Hydrocracking Process
- It is a cracking process in presence of hydrogen.
- The feedstocks are not suitable for catalytic
cracking because of their high metal, sulfur,
nitrogen, and asphaltene contents. - The process can also use feeds with high aromatic
content. - Products from hydrocracking processes lack
olefinic hydrocarbons. - The product slate ranges from light hydrocarbon
gases to gasolines to residues. - The process could be adapted for maximizing
gasoline, jet fuel, or diesel production.
29
Hydrocracking Catalysts and Reactions
- Bifunctional noble metal containing zeolites are
used. - This promote carbonium ion formation.
- Catalysts with strong acidic activity promote
isomerization. - The hydrogenation-dehydrogenation is promoted by
catalysts such as cobalt, molybdenum, tungsten,
vanadium, palladium, or rare earth elements. As
with - catalytic cracking, the main reactions occur by
carbonium ion and beta scission, yielding two
fragments that could be hydrogenated on the
catalyst surface. - The first-step is formation of a carbocation
over the catalyst surface
30
- The carbocation rearrange, eliminate a proton to
produce an olefin, or crack at a beta position to
yield an olefin and a new carbocation.
- Products from hydrocracking are saturated. i.e.
gasolines from hydrocracking units have lower
octane ratings. They have a lower aromatic
content due to high hydrogenation activity. - Products from hydrocracking units are suitable
for jet fuel use. - Hydrocracking also produces light hydrocarbon
gases (LPG) suitable as petrochemical feedstocks.
31
Hydrocracking Process
- Mostly single stage, with the possibility of two
operation modes. Once-through and a total
conversion of the fractionator bottoms by
recyling. - In once-though operation, low sulfur fuels are
produced and the fractionator bottom is not
recycled. - In the total conversion mode the fractionator
bottom is recylced to the inlet of the reactor. - In the two-stage operation, the feed is
hydrodesulfurized in the first reactor with
partial hydrocracking. Reactor effluent goes to a
high-pressure separator to separate the
hydrogen-rich gas, which is recycled and mixed
with the fresh feed. The liquid portion from the
separator is fractionated, and the bottoms of the
fractionator are sent to the second stage reactor.
32
- Hydrocracking reaction conditions vary widely,
depending on the feed and the required products.
Temperature and pressure range from 400 to 480C
and 35 to 170 atmospheres. Space velocities in
the range of 0.5 to 2.0 hr-1 are applied.
Flow diagram of a Cheveron hydocracking unit29
(1,4) reactors, (2,5) HP separators, (3) recycle
scrubber (optional), (6) LP separator, (7)
fractionator.
33
Hydrodealkylation Process
- Designed to hydrodealkylate methylbenzenes,
ethylbenzene and C9 aromatics to benzene. The
petrochemical demand for benzene is greater than
for toluene and xylenes. - After separating benzene from the reformate, the
higher aromatics are charged to a
hydrodealkylation unit. - The reaction is a hydrocracking one, where the
alkyl side chain breaks and is simultaneously
hydrogenated.
34
- Consuming hydrogen is mainly a function of the
number of benzene substituents. - Dealkylation of polysubstituted benzene increases
hydrogen - consumption and gas production (methane).
35
Hydrotreatment Processes
- Hydrotreating is a hydrogen-consuming process to
reduce or remove impurities such as sulfur,
nitrogen, and some trace metals from the feeds. - It also stabilizes the feed by saturating
olefinic compounds. - Feeds could be any petroleum fraction, from
naphtha to crude residues. - The feed is mixed with hydrogen, heated to the
proper temperature, and introduced to the reactor
containing the catalyst.
36
Hydrotreatment Catalysts and Reactions
- The same as those developed in Germany for coal
hydrogenation. - The cobalt-molybdenum/alumina is an effective
catalyst.
hydrodenitrogenation
37
Alkylation Process
- To produce large hydrocarbon molecules in the
gasoline fraction from small moleucles. (branched
hydrocarbons). - Normally acid catalyzed using H2SO4 or abhydrous
HF. - The product is known as the alkylate.
38
Some recent research has been devoted to replace
the homogeneous acid catalysts by heterogeneous
solid catalysts employing zeolites and alumina,
or zirconia.
39
Isomerization process
- Small volume but important refinery process.
- Acid catalyzed. To produce branched alkanes.
- Bifunctional catalysts activated by inorganic
chelorides are used. - Pt/zeolite is a typical isomerization catalyst.
Oligomerization of Olefines (Dimerization)
- To produce polymer gasoline with high octane
number. - Acid catalyzed. By phosphoric or sulfuric acid.
- The feed is Propylne-propane or propykene-butane
mixture. - The alkane is used as diluent.