vinod kumar cycle - PowerPoint PPT Presentation
Title:
vinod kumar cycle
Description:
about information – PowerPoint PPT presentation
Number of Views:243
Title: vinod kumar cycle
1
Cycles of Matter
- Unlike the one-way flow of energy, matter is
recycled within and between ecosystems. - These cycles are the water cycle, Nutrient Cycle,
Carbon Cycle, nitrogen cycle and phosphorus cycle.
2
Carbon Cycle
3
Water Cycle
4
Nitrogen Cycle
5
Oxygen Carbon Cycle
6
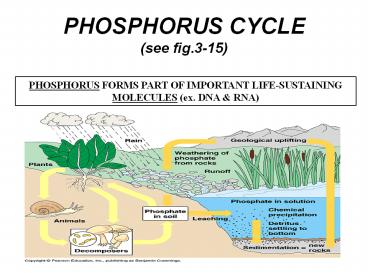
PHOSPHORUS CYCLE (see fig.3-15)
PHOSPHORUS FORMS PART OF IMPORTANT
LIFE-SUSTAINING MOLECULES (ex. DNA RNA)
7
- Phosphorus (P) is another one of the essential
elements that cycle through the ecosystem. It is
an element that is found in the ground and then
taken up by plants and animals. - Phosphorus starts its existence as phosphate
ions (PO4) in the rocks of the world. When it
rains, the phosphates and other minerals are
removed from the rocks and distributed in soils
and the water all over the planet
8
- Plants on land take in the inorganic (compounds
without carbon) phosphorus compounds from the
soil. - The phosphorus atoms are then incorporated into
many organic compounds that are used in cells. - Animals can get their phosphorus by eating plants
or drinking water. - Algae and water plants are able to absorb the
ions from the water. Unlike carbon and nitrogen,
the phosphorus cycle is not a true cycle. There
is a great deal of phosphorus lost.
9
- Phosphorus has a tendency to wind up at the
bottom of the ocean. - Once at the bottom, the phosphate ions are lost
to the world. Sometimes the phosphates are found
in runoff water and go to the bottom and
sometimes human poop contains phosphate and those
phosphates are returned to the ocean. - The problem is that all usable phosphorus
sources are on the surface The atoms are useless
once they reach the bottom of the ocean. Slowly
but surely the surface of the Earth is running
out of easy places to find phosphorus.
10
- Why is there a sudden concern? Phosphorus only
gets into the soil by the weathering process on
rocks. When plants die in the natural world, the
phosphates return to the soil. In farming, the
crops are taken away and then over many years the
soil runs out of phosphorus compounds. We have
created a situation where we must artificially
replenish the nutrients in the land. Phosphorus
is heavily used in the farming industry and
fertilizers filled with phosphates are used all
over the world to help plants grow.
11
PHOSPHORUS CYCLE (see fig.3-15)
PHOSPHORUS FORMS PART OF IMPORTANT
LIFE-SUSTAINING MOLECULES (ex. DNA RNA)
12
Phosphorus Cycle Terms
- Phosphate PO43-
- Weathering -The breakdown of rocks into smaller
rocks and soil - Sedimentation The act or process of depositing
sediment. - Geological uplift - lift up from the earth, as by
geologic forces makes mountains
13
The Carbon Cycle
14
The geological carbon cycle
- The geological component of the carbon cycle is
where it interacts with the rock cycle in the
processes of weathering and dissolution,
precipitation of minerals, burial and subduction,
and volcanism.
15
Draw the cycle on the board
- In the atmosphere, carbonic acid forms by a
reaction with atmospheric carbon dioxide (CO2)
and water. - As this weakly acidic water reaches the earth as
rain, it reacts with minerals at the earths
surface, slowly dissolving them into their
component ions through the process of chemical
weathering.
16
- These component ions are carried in surface
waters like streams and rivers eventually to the
ocean, where they precipitate out as minerals
like calcium carbonate (CaCO3). - Through continued deposition and burial, this
carbonate sediment forms the rock called
limestone (Carbonate formation).
17
- This cycle continues as seafloor spreading pushes
the seafloor under continental margins in the
process of subduction. - As seafloor carbon is pushed deeper into the
earth by tectonic forces, it heats up, eventually
melts, and can rise back up to the surface, where
it is released as CO2 and returned to the
atmosphere.
18
The Biological carbon cycle
19
The Biological carbon cycle
- Biology plays an important role in the movement
of carbon between land, ocean, and atmosphere
through the processes of photosynthesis and
respiration. - Virtually all multicellular life on Earth depends
on the production of sugars from sunlight and
carbon dioxide (photosynthesis) and the metabolic
breakdown (respiration) of those sugars to
produce the energy needed for movement, growth,
and reproduction.
20
- Plants take in carbon dioxide (CO2) from the
atmosphere during photosynthesis, and release CO2
back into the atmosphere during respiration
through the following chemical reactions - RespirationC6H12O6 (organic matter) 6O2
6CO2 6 H2O energy - Photosynthesisenergy (sunlight) 6CO2 H2O
C6H12O6 6O2
21
- Through photosynthesis, green plants use solar
energy to turn atmospheric carbon dioxide into
carbohydrates (sugars). Plants and animals use
these carbohydrates (and other products derived
from them) through a process called respiration,
the reverse of photosynthesis
22
- Respiration releases the energy contained in
sugars for use in metabolism and changes
carbohydrate fuel back into carbon dioxide,
which is in turn released to back to the
atmosphere. - Carbon dioxide is produced by all animals,
plants, fungi and microorganisms during
respiration and is used by plants during
photosynthesis
23
The Carbon Cycle
24
- Carbon dioxide is produced by all animals,
plants, fungi and microorganisms during
respiration and is used by plants during
photosynthesis - Carbon dioxide is generated as a by-product of
the combustion of fossil fuels or the burning of
vegetable matter, among other chemical processes.
- Contributes to Global warming!!!
25
Carbon Cycle Terms
- Carbon dioxide CO2 what we breathe out
- Oxygen O2 what plants produce through
photosynthesis need it to breathe - Photosynthesis plants make carbohydrates from
carbon dioxide and water
26
- Respiration - The act of inhaling and exhaling
air in order to exchange oxygen for carbon
dioxide. - Decomposition to break down decompose
- Volcanic Activity volcano releasing CO2
- Carbonate formation formation of carbonate
rocks like limestone
27
- Combustion burning fossil fuels to release
energy - Carbonate A group of minerals, all containing
the carbonate radical (CO32-)
28
View Carbon cycle animation
- carbon_cycle_version2.swf
29
THE NITROGEN CYCLE
30
The Nitrogen Cycle
- All life requires nitrogen-compounds, e.g.,
proteins and nucleic acids. - Air, which is 79 nitrogen gas (N2), is the major
reservoir of nitrogen. - But most organisms cannot use nitrogen in this
form. - Plants must secure their nitrogen in "fixed"
form, i.e., incorporated in compounds such as - nitrate ions (NO3-)
- ammonia (NH3)
- urea (NH2)2CO
- Animals secure their nitrogen (and all other)
compounds from plants (or animals that have fed
on plants).
31
- Four processes participate in the cycling of
nitrogen through the biosphere - nitrogen fixation
- decay
- nitrification
- denitrification
- Microorganisms play major roles in all four of
these.
32
- Nitrogen Fixation
- The nitrogen molecule (N2) is quite inert. To
break it apart so that its atoms can combine with
other atoms requires the input of substantial
amounts of energy. - Three processes are responsible for most of the
nitrogen fixation in the biosphere - atmospheric fixation by lightning
- biological fixation by certain microbes alone
or in a symbiotic relationship with some plants
and animals - industrial fixation
33
- Atmospheric Fixation
- The enormous energy of lightning breaks nitrogen
molecules and enables their atoms to combine with
oxygen in the air forming nitrogen oxides. These
dissolve in rain, forming nitrates, that are
carried to the earth.
34
- Biological Fixation
- The ability to fix nitrogen is found only in
certain bacteria some of which live in a
symbiotic relationship with plants or animals. - Some nitrogen-fixing bacteria live free in the
soil. - Nitrogen-fixing cyanobacteria are essential to
maintaining the fertility of semi-aquatic
environments like rice paddies.
35
Decay
- The proteins made by plants enter and pass
through food webs just as carbohydrates do. At
each trophic level, their metabolism produces
organic nitrogen compounds that return to the
environment, chiefly in excretions. The final
beneficiaries of these materials are
microorganisms of decay. They break down the
molecules in excretions and dead organisms into
ammonia.
36
Nitrification
- Ammonia (NH3)can be taken up directly by plants
usually through their roots. However, most of the
ammonia produced by decay is converted into
nitrates. This is accomplished in two steps - Bacteria oxidize Ammonia to nitrites (NO2-) which
are oxidized into nitrates (NO3-).
37
Denitrification
- Denitrification - anaerobic bacteria reduce
nitrates to nitrogen gas, thus replenishing the
atmosphere.
38
(No Transcript)
39
Nitrogen Cycle Terms
- Nitrate NO3-
- Nitrite NO2-
- Ammonium NH4
- Nitrogen Gas N2
- Nitrogen fixation -How nitrogen gets "fixed" by
bacteria to become available for plants on earth - Bacteria -are single-celled microorganisms
- Lightning - electrical storms
40
- Nitrification - the biological oxidation of
ammonia with oxygen into nitrite followed by the
oxidation of these nitrites into nitrates - Denitrification- bacteria break down nitrates to
give nitrogen gas, which returns to the
atmosphere - Decomposition breaking down dead material
41
Nitrogen cycle animation
- ncycle1.swf