Additions to Alkenes - PowerPoint PPT Presentation
1 / 82
Title:
Additions to Alkenes
Description:
ketone. 58. Reaction with both esters and acyl halides. Two ... OH, -NH2, -NHR, -CO2H, -SO3H, -SH, -CC-H, aldehyde, ketone, esters, amide -NO2, -CN, epoxide ... – PowerPoint PPT presentation
Number of Views:486
Avg rating:3.0/5.0
Title: Additions to Alkenes
1
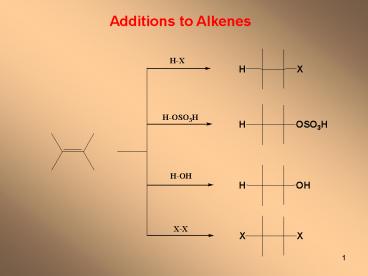
Additions to Alkenes
2
Additions to Alkenes
Reactions usually hexothermic
Electron-rich region
Susceptible of electrophilic attack
Electron-rich region
3
Electrophiles are Lewis Acids
4
Markovnikovs rule
Observation
C atom with the greater of H
5
Markovnikovs rule
Explanation
t-butyl bromide !!
6
(No Transcript)
7
General formulation In the ionic addition of an
unsymmetrical reagent to a double bond, the
positive portion of the adding reagent attaches
in the way that generates the most stable
carbocation.
These reactions are called regioselective
8
Stereochemistry of the addition reaction
Addition of H2SO4 to form the alkyl hydrogen
sulphate X HSO4 Addition of H2O to form
the corresponding alcohol X OH (it
needs acid as a catalyst. Carbocation
transpositions are possible with 10 and 20
carbocations)
A valuable reaction
9
Addition of halogens
Fast, high yield reactions forming vic-dihalides
10
Mechanism
anti addition a stereospecific reaction
11
Stereospecificity
The reaction forms only one steroisomeric form
(trans) but is not enantiospecific
12
Stereospecificity
(MODELS)
13
Stereospecificity
R
S
14
Formation of halohydrins (addition of X2 and H2O)
Use excess of water to favor halohydrin
15
Formation of halohydrins (addition of X2 and H2O)
mechanism
anti attack
16
Cyclopropanation reactions (addition of carbenes)
diazomethane
CH2 N2
HCCl3 RO- RO-H
CCl2 Cl-
Carbenes are highly unstable (in situ generated)
17
Cyclopropanation reactions (addition of carbenes)
18
Alkene Oxidations
Syn-hydroxylation
OH-, H2O
CH2CH2 KMnO4 H2C CH2
OH OH
Two methods
Na2SO3/H2O
CH2CH2 OsO4
H2C CH2
OH OH
19
Mechanism
20
Oxidative cleavage
21
Ozonolysis
22
Synthesis of alkynes
23
Synthesis of alkynes
Prepare the alkene (large variety of preparation
methods enables a variety of molecular
architectures) Destroy the double bond by
adding Br2 thus generating a vic-dibromide Doubl
e elimination reaction performed with NaNH2
generates the alkyne No stereochemical
implications gem-dihalides may be used as well
24
The acidity of terminal alkynes
sp2
sp
sp3
pKa 25
44 50
More s character, more stable anion, weaker
conjugated base, better acid
Acidity CH3CH3 lt CH2CH2
lt HCCH Basicity CH3CH2 gt
CH2CH gt HCC
OH lt OR lt CCR lt NH2 lt CHCH2 lt CH2-CH3
25
Alkynides as Good Nucleophiles for
SN2 Good Base for E2
26
Alkynes on the same line as alkenes
Addition of Br2 and Cl2 Addition of
HX Oxidative cleavage
27
Hydrogenation of alkynes
Syn addition for the formation of cis-alkenes
Anti addition for the formation of trans-alkenes
28
Anti addition the mechanism
29
Chemistry of Alcohols and ethers Chapter 11
30
Nomenclature
Just add the suffix ol. All the other IUPAC
rules still apply CH3OH CH3CH2OH
CH3CH(OH)CH3 CH3C(CH)3(OH)CH3 Methan
ol Ethanol 2-propanol
2-methyl-2-propanol
(Isopropanol)
(t-butanol) 10 alcohol 10
20
30
Common names
Phenol benzyl alcohol
2-propenol 2 propynol
31
Examples Give the IUPAC names to the following
2,4-dimethyl-1-pentanol
4-phenyl-2-pentanol
4-pentene-2-ol
32
Ethers Nomenclature
R-O-R dialkyl ether CH3OCH2CH3
CH3CH2OCH2CH3
C6H5OC(CH3)3 Ethyl methyl ether
diethyl ether t-butyl phenyl
ether
Just for complex and polyfunctional
ethers CH3CHCH2CH2CH3
CH3OCH2CH2OCH3
OCH3
2-methoxypentane 1-ethoxy-4-methylbenze
ne 1,2-dimethoxyethane
33
Physical properties
Alcohols much higher BP and MP with
respect to the corresponding
hydrocarbons (hydrogen bridges are very
strong) Ether very comparable to
the corresponding hydrocarbon
(Et2O bp. similar to pentane)
34
Synthesis of alcohols
1. acid-catalyzed hydration of alkenes
(follows Markovnikovs rule)
Markovnikov rule the bulkier group goes on the
most crowded C atom
35
Carbocation rearrangements
36
(No Transcript)
37
(No Transcript)
38
Synthesis of alcohols
2. Oxymercuration-demercuration (also follows
Markovnikovs rule but no skeleton
rearrangement)
39
Mechanism
1st step Hg(OAc)2
Hg(OAc) OAc- 2nd
step 3rd step 4th step
40
The reaction can also be used for the synthesis
of ethers just using ROH instead of H2O
41
3. Addition of water to the double bond
(anti-Markovnikov)
1st step Hydroboration
Less crowded C atom
42
3. Addition of water to the double bond
anti-Markovnikov
2nd step oxidation / hydrolysis
NaOH
R3B H2O2 3 R-OH
Na3BO3
43
- Summary of Stereochemistry of hydroboration
- Anti-markovnikov addition of H2O to the olefin
- 2. Syn addition
44
The basic principle of asymmetric synthesis
S,R S,S
diastereomers !!! (different
stability and rate of
formation)
Enantiomeric enrichment
45
4. Alcohols from ketones and aldehydes
d -d
- sp2
- trigonal planar (1200)
- strongly polarized double bond
Reactivity?
addition
nucleophilic
46
Nucleophilic Addition
- Change of hybridation
- Adding complexity to the molecular structure
- Converting a carbonyl to alcohol
47
From the qualitative point of view
Oxidation
Reduction Gaining O (increase ox
state) Loosing O (decrease ox
) Loosing H
Gaining H
RCH2OH RCH3
reduction RCH2OH
RC(O)OH oxidation
48
Aldehyde
alcohol H2 LiAlH4 NaBH4
Cr2O72- MnO4- OsO4 Ce4
49
Mechanism of reduction
Ease of reduction
50
5. Using Organometallic compounds (Chemistry of
carbanions)
M Na, K Mg, Li
Pb, Sn, Hg, Tl
Versatile and useful
explosive
strong base (H-abstraction), strong nucleophiles
51
Preparation
R Br 2 Li R Li
LiBr
Ethers are the preferred solvents but
ethers are slowly attacked and decomposed
52
Grignard reagents the most popular
- possible with a great variety of R
- ethers as solvent are crucial to the success of
the reaction
53
-d d
R - M M Mg, Li
The strong polarity of the M-C bond and the
consequent carbanionic character determines the
reactivity
1) superstrong base
54
-d d
R - M M Mg, Li
The strong polarity of the M-C bond and the
consequernt carbanionic character determines the
reactivity
2) very good nucleophiles
55
and of course E2 reactions
56
Nucleophilic Addition (prep. of ROH)
Use acid hydrolysis to avoid precipitation of
Mg(OH)2
57
Formation of alcohols
Formaldehyde Aldehyde ketone
58
Reaction with both esters and acyl halides
Two identical groups
59
Planning a Grignard Synthesis
We can synthesize ANY alcohol
60
Restrictions
Incompatible with any function that can be
deprotonated -OH, -NH2, -NHR, -CO2H, -SO3H,
-SH, -CC-H, aldehyde, ketone, esters,
amide -NO2, -CN, epoxide
Grignard carbonatation to lenght the chain by one
C atom
61
Reactivity of alcohols
Basic center
SN2
62
Acidity
CH3OH more acidic than H2O (just a
little) Crowded ROH less acidic than H2O
(solvation of the conjugated base RO-)
Acidity H2O gt ROH gt RCCH gt H2 gt NH3 gtgtgt
RH Basicity R- gtgtgt NH2- gt H- gt RCC- gt RO- gt OH-
Useful reagents in organic synthesis (E and SN
reactions)
63
Esterification
Ester of alkylsulfonic acid
ester
64
Mechanism of formation
65
Excellent leaving groups for SN2 reactions
66
Two-step substitution
67
Conversion into R-X
SN1 reaction HI gt HBr gt HCl gt HF 30 gt 20 gt 10 lt
Me Acid catalyzed
SN2-type
E-products unavoidable Rearrangements
68
A trick to enhance SN2 and to avoid elimination
and rearrangements
69
Use of PBr3
- Only for 10 and 20 without rearrangement
70
Use of thionyl chloride
No skeletal rearrangements !!
71
Synthesis of Ethers
Intermolecular dehydration of alcohols
H2SO4
R-OH HO-R R-O-R
H2O alkene
Step 1 Step 2 Step 3
72
Williamson synthesis R-ONa R L
R O R
NaL - L must be a good leaving group
(tosylate, Br, I) - Versatile for the synthesis
of asymmetric ethers - Usual limitation of SN2
reactions
73
Using reversible formation of ethers to protect
OH functions
Example HOCH2CH2CH2Br
NaCCH NaOCH2CH2CH2Br
HCCH But HOCH2CH2CH2Br
(CH3)3COCH2CH2CH2Br
(CH3)3COCH2CH2CH2Br NaCCH
(CH3)3COCH2CH2CH2CCH
(CH3)3COCH2CH2CH2CCH
HOCH2CH2CH2CCH
74
As an alternative
75
Ether Cleavage
Very strong acid are needed
76
Epoxides
Lets do the following experiment
Epoxide (oxyrane)
77
Reactions of epoxides
Ring opening with acid with base
78
Stereochemistry of ring opening
Acid catalyzed
Resembles a 30 carbocation
(S)
(R)
Base catalyzed
(S)
(S)
79
Anti hydroxylation of alkenes
80
Stereochemistry
meso
cis
81
racemate
trans
82
(No Transcript)