I. Spectroscopy - PowerPoint PPT Presentation
1 / 18
Title:
I. Spectroscopy
Description:
I. Spectroscopy. Study of the interaction of electromagnetic ... not just visible light!! http://www.dnr.sc.gov/ael/personals/pjpb/lecture/spectrum.gif ... – PowerPoint PPT presentation
Number of Views:337
Avg rating:5.0/5.0
Title: I. Spectroscopy
1
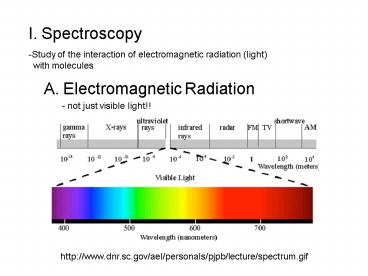
I. Spectroscopy
- Study of the interaction of electromagnetic
radiation (light) - with molecules
A. Electromagnetic Radiation
- not just visible light!!
http//www.dnr.sc.gov/ael/personals/pjpb/lecture/s
pectrum.gif
2
1. Wave Behavior of Light
a. Wavelength (?)
- often given in cm or nm
b. Frequency (?)
- of wave cycles that pass a point in a second
- s-1
3
2. Particle Behavior
a. Photon massless packet of energy
b. A little bit of math!
- First c ??, where c is the speed of light
- (3.00 x 108 m s-1)
- ? h? , where h is Plancks Constant
- since we know that ? c/?
- then we also know that ? hc/?
- So??
- this means that the energy of the light is
directly - proportional to the frequency
- energy of light is inversely proportional to
wavelength
4
3. Absorption
- light of particular energy can be absorbed by a
molecule - resulting in the molecule adopting an excited
state - - this absorption can be measured!!
5
B. Infrared (IR) Spectroscopy
- Upon absorption of light in the infrared region
of the - electromagnetic spectrum by organic molecules
- results in molecular motion
1. Types of Motion
6
2. What we observe
- data is recorded using wavenumbers ( ) where
- 1/?, (cm-1)
- this means energy is directly proportional to
- wavenumber, i.e. higher wavenumber, higher
energy - can think of as weights on springs
a. Effect of Atom Type
C(sp3) H 2960 - 2850 cm-1 O
H (alcohol) 3600 - 3200 cm-1
b. Effect of Bond Strength
CC 1680 - 1620 cm-1 CC 2260 - 2100 cm-1
7
3. Sample Spectrum
8
4. Table of IR Data
9
5. Trends
a. Hydrogen Region 3330 - 2700 cm-1
O H(alc) 3650 - 3200 cm-1 O H(CA) 3300 -
2500 cm-1 N H(amine) 3500 - 3250 cm-1 N
H(amide) 3500 - 3400 cm-1
C(sp3) H 2960 - 2850 cm-1 C(sp2) H 3150 -
3050 cm-1 C(sp) H 3300 cm-1
b. Triple Bond Region 2300 - 2000 cm-1
CC(alkyne) 2260 - 2100 cm-1 CN(nitrile)
2260 - 2220 cm-1
c. Double Bond Region 1900 - 1500 cm-1
C O(ald) 1740 - 1690 cm-1 C O(ket) 1750 -
1680 cm-1 C O(est) 1750 - 1735 cm-1
C O(CA) 1760 - 1710 cm-1 C O(am) 1690 - 1630
cm-1 C C 1680 - 1620 cm-1
10
5. Trends
d. Fingerprint Region
- Not much information, except!! C(sp2, aromatic,
bending) H Mono-substituted 770 - 730, 720 -
680 cm-1 Di-substituted (ortho) 770 - 735
cm-1 Di-substituted (meta) 810 - 750
cm-1 Di-substituted (para) 860 - 800 cm-1
11
II. Sample Spectra
A. Spectrum 1
- strong versus medium versus weak Why?
- Dipole moment of molecule must change as
vibration occurs - Number of bonds absorbing
12
B. Spectrum 2
- Sharp versus Broad Signals Why?
- multiple rotational energy levels in each
vibrational energy level - ??
13
Multiple Rotational Energy Levels??
14
C. Spectrum 3 What functional groups are
present?
15
D. Spectrum 4 What functional groups are
present?
16
E. Spectrum 5 What functional groups are
present?
17
F. Spectrum 6 Propose a structure for
a compound with MF C7H12O
18
G. Spectrum 7 Propose a structure for
a compound with MF C5H12O